Key Takeaways
This systematic review and meta-analysis, published in JAAD Reviews in 2025, pooled data from six randomized controlled trials involving 1,104 men with androgenetic alopecia (male pattern hair loss) to assess the safety of topical finasteride. The researchers found no statistically significant increase in dermatologic side effects (such as contact dermatitis, pruritus, or erythema) or sexual dysfunction side effects (erectile dysfunction and decreased libido) compared to placebo. No cases of depression were reported among topical finasteride users. The findings suggest a favorable safety profile, but the authors are transparent about important limitations: sample sizes were small, follow-up periods averaged only about 24 weeks, the studies tracked treatment-emergent adverse events that may not have been caused by the treatment, and rare side effects could easily go undetected. Most topical finasteride in clinical practice is compounded with variable formulations, which introduces additional uncertainty not fully captured by these trials.
Context and Purpose
Finasteride is one of the most studied treatments for male pattern hair loss. In its oral form (1 mg daily), it has been FDA-approved since 1997. It works by inhibiting 5-alpha-reductase, the enzyme that converts testosterone to dihydrotestosterone (DHT), which is the primary androgen responsible for shrinking hair follicles in genetically susceptible individuals.
In recent years, topical finasteride has emerged as an alternative delivery method, with the hope that applying finasteride directly to the scalp could reduce DHT locally while limiting systemic exposure and, by extension, systemic side effects. However, topical finasteride is not FDA-approved. In fact, the FDA issued a public alert cautioning both healthcare providers and consumers about potential risks, citing 32 adverse event reports filed between 2019 and 2024 through the FDA Adverse Event Reporting System. These reports included erectile dysfunction, depression, and skin reactions, with some individuals reporting persistent symptoms after stopping use.
This created a tension: a growing number of clinicians and patients were turning to topical finasteride, yet the best available evidence on its safety had not been systematically compiled. The authors of this study set out to fill that gap by analyzing only randomized, placebo-controlled trials, the gold standard for evaluating treatment effects.
How the Research Was Conducted
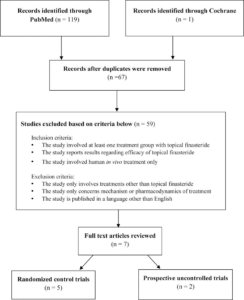
The team followed PRISMA guidelines, a widely accepted protocol for transparent systematic reviews. They searched three major medical databases (MEDLINE, Embase, and CENTRAL) for randomized controlled trials comparing topical finasteride to placebo in men with androgenetic alopecia.
Two independent reviewers screened the articles, ultimately identifying six studies that met inclusion criteria. These six trials collectively enrolled 1,104 male patients with a mean age of 32.7 years (range: 27 to 49). The average treatment duration across studies was approximately 23 to 24 weeks.
The researchers used a random-effects meta-analysis to pool results, calculating risk ratios (RR) with 95% confidence intervals. A risk ratio above 1.0 would suggest a higher likelihood of side effects with topical finasteride compared to placebo, while a value below 1.0 would suggest a lower likelihood. They also measured statistical heterogeneity using the I² statistic, which indicates how much variation across studies is due to genuine differences rather than chance.
Results
Among the 1,104 participants, 139 patients (12.6%) experienced at least one adverse event. Of those:
- 86.3% (120 of 139) had dermatologic adverse events
- 12.9% (18 of 139) had sexual dysfunction adverse events
- 0.7% (1 of 139) had a mood change adverse event
Dermatologic Side Effects
None of the dermatologic outcomes reached statistical significance compared to placebo:
- Alopecia (hair shedding): RR = 0.83 (95% CI: 0.38–1.80)
- Contact dermatitis: RR = 0.66 (95% CI: 0.27–1.64)
- Application site erythema (redness): RR = 3.09 (95% CI: 0.46–21.02)
- Pruritus (itching): RR = 1.47 (95% CI: 0.85–2.52)
- Seborrheic dermatitis: RR = 0.74 (95% CI: 0.05–10.46)
It is worth noting that application site erythema had the highest point estimate (RR of 3.09), suggesting a possible trend, but the confidence interval was extremely wide, crossing 1.0, meaning the finding was not statistically significant. This wide range reflects uncertainty from small sample sizes.
Sexual Dysfunction
Neither erectile dysfunction nor decreased libido reached statistical significance:
- Erectile dysfunction: RR = 1.36 (95% CI: 0.26–7.11)
- Decreased libido: RR = 0.27 (95% CI: 0.07–1.08)
The decreased libido finding is notable: the point estimate actually favored topical finasteride over placebo (RR below 1.0), though it just barely missed statistical significance. Heterogeneity for both sexual dysfunction outcomes was 0%, meaning the studies were remarkably consistent.
Mood Changes
No cases of depression were reported in any patient receiving topical finasteride across all six trials.
Conclusions
The authors conclude that topical finasteride exhibits a “favorable safety profile” based on the available randomized controlled trial data. They note that a separate pharmacovigilance study of FDA data similarly found fewer sexual and neuropsychiatric adverse event signals with topical versus oral finasteride, which aligns with their findings.
However, there are important caveats that deserve honest attention:
- Small sample size: With only 1,104 patients across six trials, this analysis lacks the statistical power to detect rare adverse events. A side effect occurring in 1 in 500 users, for example, could easily go unnoticed.
- Short follow-up: The average study duration was roughly 24 weeks. We know very little about the safety profile beyond six months, let alone across years of continuous use.
- Treatment-emergent vs. treatment-caused: The trials tracked adverse events that appeared during treatment, but correlation is not causation. Some of these events may have occurred regardless of treatment.
- Compounding variability: Most topical finasteride in real-world use is compounded by pharmacies, with variable concentrations, vehicles, and penetration profiles. The standardized formulations used in clinical trials may not reflect what most patients actually receive.
- Wide confidence intervals: Several outcomes had confidence intervals so wide that both meaningful harm and meaningful benefit remain plausible. Absence of statistical significance is not the same as proof of safety.
Relevance to Topical Alopecia Treatments
This meta-analysis is directly relevant to anyone interested in or currently using topical hair loss solutions. The central question it addresses, whether applying finasteride to the scalp avoids the systemic side effects associated with oral finasteride, is one of the most common concerns among individuals considering treatment.
Pharmacokinetic studies have shown that topical finasteride does reduce plasma DHT, but to a lesser degree than the oral form. This meta-analysis provides clinical evidence consistent with that pharmacokinetic rationale: if less finasteride reaches the bloodstream, one would expect fewer systemic side effects. The data here support that hypothesis, though they do not prove it conclusively.
For topical formulation science, the authors’ note about compounding variability is particularly important. The safety data in these trials came from controlled, standardized preparations. The growing market for compounded topical finasteride, often combined with minoxidil or other actives, introduces variables in concentration, absorption, and stability that may alter both efficacy and safety in ways these trials cannot address.
Who This Study Matters Most For
This research is most relevant to men with androgenetic alopecia who are weighing the potential benefits and risks of topical finasteride, particularly those who have considered oral finasteride but are concerned about sexual or mood-related side effects. It offers reassurance, though not certainty, that the topical route may carry a lower risk of these specific adverse events.
It also matters for clinicians prescribing topical finasteride, providing them with the strongest level of evidence currently available, pooled randomized controlled trial data, to guide conversations with patients. And it matters for regulators and compounding pharmacies, highlighting the need for standardized formulations and continued surveillance.
For individuals already using topical finasteride without issues, this study provides context for their experience. For those still deciding, it is one important piece of a larger puzzle that includes efficacy data, individual risk factors, and honest conversations with a dermatologist.
Sources and Citations
Primary Study
Bai JQA, Geng RSQ, Donovan J. Adverse events of topical finasteride for male androgenetic alopecia: A systematic review and meta-analysis of randomized controlled trials. JAAD Reviews. 2025;6:72-74. doi:10.1016/j.jdrv.2025.09.003
Selected References Cited by the Authors
- Lolli F, Pallotti F, Rossi A, et al. Androgenetic alopecia: a review. Endocrine. 2017;57(1):9-17. doi:10.1007/s12020-017-1280-y
- Center for Drug Evaluation and Research. FDA alerts health care providers, compounders and consumers. U.S. Food and Drug Administration. https://www.fda.gov/drugs/human-drug-compounding/fda-alerts-health-care-providers-compounders-and-consumers-potential-risks-associated-compounded
- Caserini M, Radicioni M, Leuratti C, et al. Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men with androgenetic alopecia. Int J Clin Pharmacol Ther. 2016;54(1):19-27. doi:10.5414/CP202467
- Pozo-Perez L, Tornero-Esteban P, Lopez-Bran E. Clinical and preclinical approach in AGA treatment: a review of current and new therapies in the regenerative field. Stem Cell Res Ther. 2024;15(1):260. doi:10.1186/s13287-024-03801-5
- Gupta AK, Talukder M, Keene SA, Bamimore MA. Is the safety of finasteride correlated with its route of administration: topical versus oral? A pharmacovigilance study. Int J Dermatol. Published online July 15, 2025. doi:10.1111/ijd.17957