Key Takeaways
This systematic review, published in the Journal of Drugs in Dermatology, examined seven clinical studies on topical finasteride for the treatment of androgenetic alopecia (pattern hair loss) in men and women. Across all included studies, topical finasteride was associated with a significant decrease in the rate of hair loss, increases in total and terminal hair counts, and positive hair growth assessments. Critically, topical application reduced scalp and plasma DHT, the hormone most implicated in follicular miniaturization, without significantly altering serum testosterone levels. Reported side effects were mild and uncommon. The combination of topical finasteride with minoxidil appeared more effective than either agent alone. However, the evidence base remains small: only 256 total subjects were studied across all seven papers, most studies were short in duration, and no large-scale, long-term trials had been completed at the time of review. These results are encouraging but far from definitive.
Context and Purpose
Androgenetic alopecia affects millions of men and women worldwide. It is driven by dihydrotestosterone (DHT), a potent androgen that binds to receptors in the hair follicle and progressively shrinks it. Over time, thick terminal hairs become fine vellus hairs, and eventually production may cease altogether.
For decades, the only FDA-approved pharmacologic options have been oral finasteride (for men) and topical minoxidil (for men and women). Oral finasteride works well for many patients, blocking the enzyme that converts testosterone to DHT. But it carries a side-effect profile that gives many patients pause: sexual dysfunction, breast tenderness, and mood changes have all been reported. Women of childbearing age cannot take it at all, as it is classified as a Category X drug in pregnancy.
The question that motivated this review is a straightforward one: can finasteride be delivered through the skin of the scalp, targeting the follicle directly, while sparing the rest of the body from unwanted hormonal effects? Researchers from the University of California, Irvine, set out to gather and evaluate every published clinical study that had tested this idea in humans.
How the Research Was Conducted
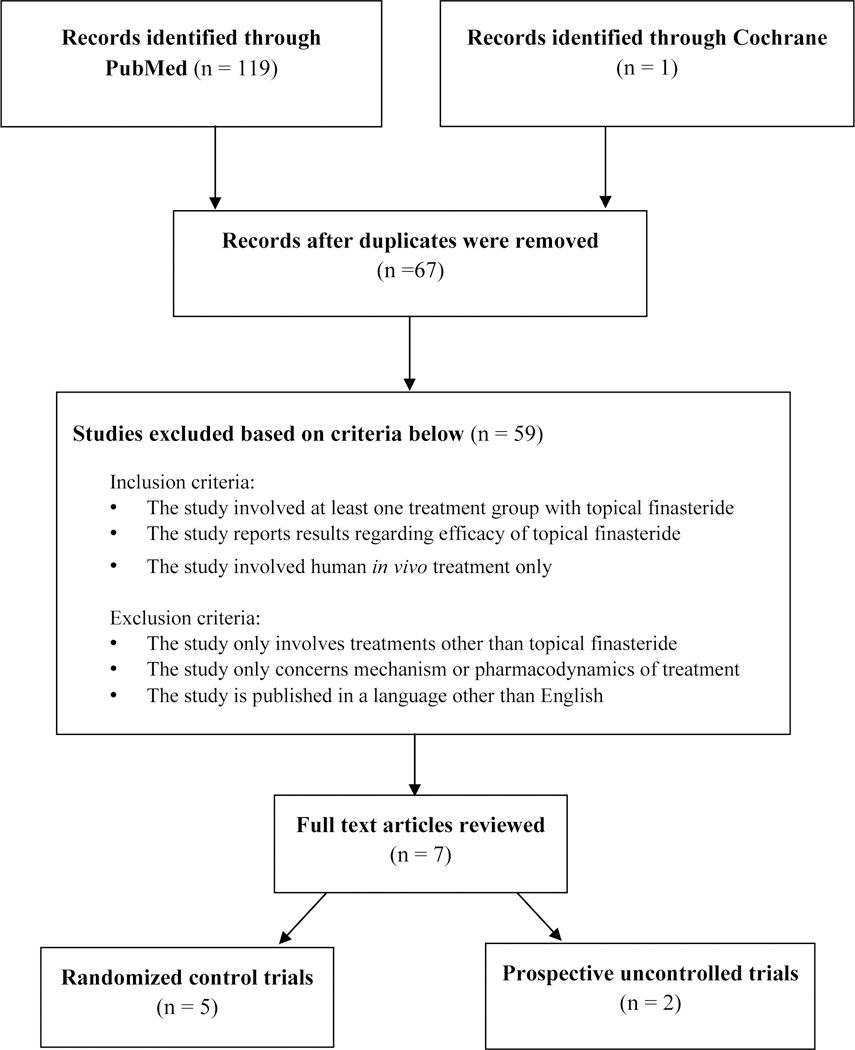
The authors searched PubMed/MEDLINE, Embase, PsycINFO, TRIP, the Cochrane Library, and the Cochrane Skin databases using search terms including “topical finasteride,” “finasteride solution,” “finasteride liquid,” “finasteride foam,” and “finasteride cream.” Only studies of topical finasteride used on living human subjects were included. Studies had to report at least one treatment group using topical finasteride and provide efficacy results.
From an initial pool of 119 articles, 67 were identified by title. After removing duplicates and applying inclusion criteria, seven studies remained: five randomized controlled trials and two prospective studies. A total of 256 subjects (232 male, 24 female) participated across all studies.
Results
Efficacy of Topical Finasteride Alone
The earliest study, by Mazarella et al. in 1997, tested a very low concentration of topical finasteride (0.005%) applied twice daily for 16 months. Even at this low dose, there was a significant decrease in the rate of hair loss after six months. Seventy-three percent of treated patients reported “high effectiveness,” compared to 60% of placebo patients reporting “no effect.”
A later randomized trial by Hajheydari et al. compared 1% topical finasteride gel to oral finasteride 1 mg in 38 men. Both groups showed statistically significant increases in total and terminal hair count after four months, with no significant difference between the two delivery methods. Notably, the topical group saw increased terminal hair counts by month three.
Pharmacodynamic Findings
Two studies by Caserini et al. provided particularly valuable pharmacodynamic data. In the first, topical finasteride 0.25% applied twice daily reduced plasma DHT by 68-75%, compared to a 62-72% reduction with oral finasteride. Importantly, neither route of administration significantly altered plasma testosterone.
In a follow-up dose-finding study, a surprising result emerged: once-daily application of topical finasteride was more effective at reducing scalp DHT than twice-daily application. The authors concluded that lower volumes (100 or 200 microliters) applied once daily may be the ideal regimen, as higher doses (300-400 microliters) increased plasma finasteride levels without additional scalp benefit, potentially raising systemic risk.
Combination Therapies
Several studies examined topical finasteride combined with other agents. A randomized, double-blind trial by Tanglertsampan compared topical minoxidil 3% alone to a combination of minoxidil 3% with finasteride 0.1%. Both groups showed increased hair counts, but the combination group achieved statistically superior improvement.
Rafi and Katz tested a novel combination of topical finasteride, dutasteride, and minoxidil in 15 men. All patients showed significant hair growth. Those who added oral finasteride, topical minoxidil foam, and ketoconazole shampoo experienced visible growth in as little as 30 days, while those using the topical combination alone saw significant growth after three months.
Chandrashekar et al. followed 50 men who had been on oral finasteride and topical minoxidil for two years and then switched to a topical minoxidil 5% and finasteride 0.1% combination. Approximately 80-84% of patients maintained good hair density on the topical-only regimen, even those who had a treatment gap of 8-12 months before switching.
Safety and Side Effects
Across all seven studies, serious adverse events were absent. Reported side effects were mild and infrequent:
- Scalp erythema and contact dermatitis (in a small number of patients)
- Increased liver enzymes (alanine aminotransferase) in two participants
- Testicular pain, headache, presyncope, conjunctivitis, and oropharyngeal pain, each reported rarely
No changes in serum testosterone were observed in any study. Patient compliance and satisfaction with topical finasteride were generally high.
Conclusions
The cumulative evidence from these seven studies suggests that topical finasteride can reduce scalp DHT, slow hair loss, and promote measurable hair regrowth, with a side-effect profile that appears milder than that of oral finasteride. The data also suggest that topical finasteride is non-inferior to oral finasteride for hair regrowth.
But it is essential to be honest about what the evidence does not prove. The total number of subjects studied is small, 256 people across all trials. Most studies were short, ranging from seven days (pharmacodynamic studies) to 16 months. There were no large, multi-center, long-term randomized controlled trials. Vehicle formulation varied across studies (gel versus solution), and no head-to-head comparisons of delivery systems were conducted. The optimal concentration, volume, and frequency of application remain uncertain, though once-daily application of low-volume 0.25% solution appears promising.
Furthermore, only 24 of the 256 subjects were women, making it difficult to draw firm conclusions about topical finasteride’s use in female pattern hair loss, despite the theoretical appeal of a topical route that might avoid the teratogenic risks of systemic exposure.
Relevance to Topical Alopecia Treatments
This review carries direct implications for anyone interested in topical approaches to treating hair loss. The central finding, that finasteride can be delivered through the scalp with measurable DHT suppression and reduced systemic exposure, validates the broader concept that topical formulations of known anti-androgenic agents deserve serious investigation.
Several specific points stand out for the topical treatment landscape:
- Combination is likely superior to monotherapy. Studies consistently showed that pairing topical finasteride with minoxidil produced better outcomes than either agent alone. This supports the rationale behind multi-ingredient topical formulations.
- Vehicle and delivery matter enormously. The review highlights that nanoparticle delivery, liposomes, and absorption enhancers such as ethanol and propylene glycol can improve finasteride’s penetration into the dermis. The formulation science behind a topical product may be as important as the active ingredient itself.
- Less may be more. Lower application volumes once daily outperformed higher volumes or twice-daily regimens in reducing scalp DHT. This challenges the intuition that more frequent application means better results, and suggests that optimizing delivery is more important than maximizing dose.
- A potential path for women. While the evidence in women is extremely limited, topical finasteride’s reduced systemic absorption may eventually open a treatment avenue that systemic finasteride cannot offer to female patients.
Who This Study Matters Most For
This review is most relevant for men with androgenetic alopecia who are reluctant to take oral finasteride due to concerns about sexual side effects, mood changes, or other systemic risks. It provides early evidence that a topical route may offer comparable benefits with a more favorable safety profile.
It also matters for women with pattern hair loss, a population that has had even fewer pharmacologic options than men. While the data in women remain sparse, the theoretical advantage of topical delivery, reducing systemic hormonal suppression, makes this an area worth watching closely.
Patients who have previously responded to oral finasteride and minoxidil but wish to simplify or de-escalate their regimen may find the switch to a combined topical formulation viable, as the Chandrashekar study suggests that hair density can be maintained after such a transition.
Finally, clinicians and researchers working on next-generation topical formulations will find this review useful as a benchmark: it maps the current state of evidence and clearly identifies the gaps, from optimal vehicle design to long-term safety data, that future studies must fill.
Sources and Citations
Primary Study
Lee SW, Juhasz M, Mobasher P, Ekelem C, Atanaskova Mesinkovska N. A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. J Drugs Dermatol. 2018;17(4):457-463.
Selected References Cited by the Authors
- Mazarella GF, Loconsole GF, Cammisa GA, et al. Topical finasteride in the treatment of androgenic alopecia. Preliminary evaluations after a 16-month therapy course. J Dermatol Treat. 1997;8(3):189-192.
- Hajheydari Z, Akbari J, Saeedi M, Shokoohi L. Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. Exp Gerontol. 2002;37(9-10):981-990.
- Caserini M, Radicioni M, Leuratti C, et al. A novel finasteride 0.25% topical solution for androgenetic alopecia: pharmacokinetics and effects on plasma androgen levels. Int J Clin Pharmacol Ther. 2014;52(10):842-849.
- Caserini M, Radicioni M, Leuratti C, et al. Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men with androgenetic alopecia. Int J Clin Pharmacol Ther. 2016;54(1):19-27.
- Tanglertsampan C. Efficacy and safety of 3% minoxidil versus combined 3% minoxidil/0.1% finasteride in male pattern hair loss. J Med Assoc Thai. 2012;95(10):1312-1316.
- Chandrashekar BS, Nandhini T, Sriram R, Navale S. Topical minoxidil fortified with finasteride: An account of maintenance of hair density after replacing oral finasteride. Indian Dermatol Online J. 2015;6(1):17-20.
- Rafi AW, Katz RM. Pilot study of 15 patients receiving a new treatment regimen for androgenic alopecia: the effects of atopy on AGA. ISRN Dermatol. 2011;2011:241953.