Key Takeaways
- What was studied: A systematic review of nine studies compared dutasteride and finasteride, two oral 5-alpha reductase inhibitors, in treating androgenetic alopecia (AGA) in men and women.
- What was found: Dutasteride at 0.5 mg daily was significantly more effective than finasteride at 1 mg daily in increasing hair count and hair thickness. All active treatment groups outperformed placebo. Adverse event profiles were comparable between the two drugs.
- Why it matters: For people experiencing pattern hair loss who do not respond adequately to finasteride, dutasteride may represent a stronger pharmacological option, though it remains off-label for AGA in most countries.
- Limitations: Only nine studies were included, seven enrolled only men, and the two female-focused studies were relatively small. No large, high-quality randomized trial has specifically examined dutasteride for female AGA. Long-term safety data remain limited.
Context and Purpose
Androgenetic alopecia is the most common form of hair loss worldwide. It affects roughly half of Caucasian men and nearly one in five Caucasian women, with prevalence climbing as people age. The condition is driven by dihydrotestosterone (DHT), a potent androgen that shrinks hair follicles through a process called miniaturization, gradually transforming thick terminal hairs into fine, barely visible vellus hairs.
Two classes of oral medication target this pathway. Finasteride blocks the type 2 isoform of the enzyme 5-alpha reductase, reducing serum DHT by about 70%. Dutasteride blocks both type 1 and type 2 isoforms, reducing DHT by roughly 90%. Finasteride at 1 mg daily is FDA-approved for male pattern hair loss. Dutasteride, approved for benign prostatic hyperplasia, is not FDA-approved for hair loss, yet clinicians prescribe it off-label with increasing frequency.
This systematic review, published in Dermatology Reports in 2024 by researchers from King Saud University and collaborating institutions in Saudi Arabia, set out to directly compare the two drugs in terms of efficacy, safety, and side effects across both sexes.
How the Research Was Conducted
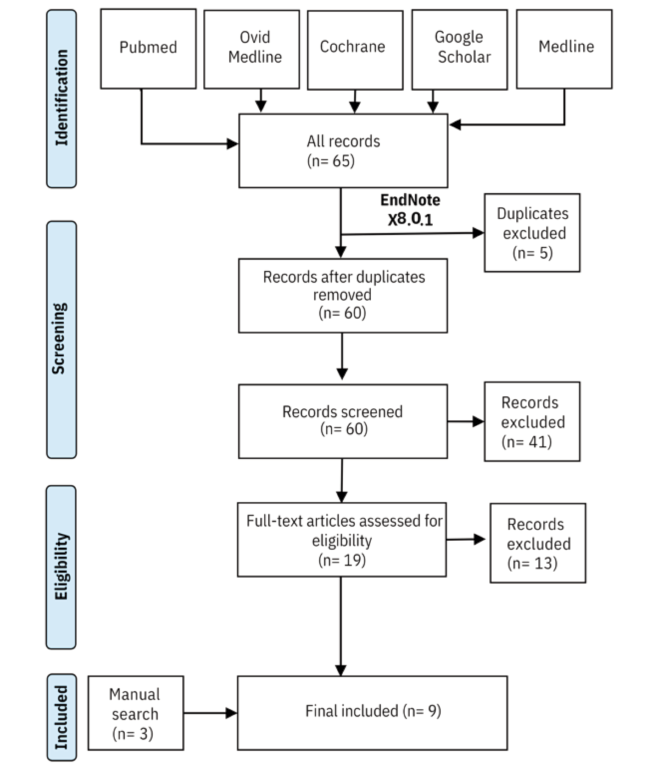
The authors searched PubMed, Ovid Medline, Google Scholar, and the Cochrane Library using terms related to dutasteride, finasteride, pattern hair loss, efficacy, tolerability, and side effects. They included any study reporting on the safety and efficacy of either drug for AGA, regardless of dose, sex, or route of administration, but excluded case reports, review articles, and conference abstracts.
After screening 60 records by title and abstract, then evaluating 19 full-text articles, and supplementing with manual searches, the team arrived at nine studies for final analysis:
- Four randomized controlled trials (RCTs)
- One single-arm clinical trial
- Two prospective cohort studies
- Two retrospective cohort studies
Seven of the nine studies enrolled only male participants. The two female-focused studies were a prospective cohort using dutasteride mesotherapy and a retrospective cohort comparing low-dose dutasteride with finasteride. Study quality was assessed using the National Institutes of Health quality assessment tool.
Results
Hair Count
Six studies reported hair count outcomes, and the pattern was consistent: all active treatment groups showed statistically significant increases in hair count compared to placebo. The key comparisons included:
- In Eun et al.’s RCT, dutasteride 0.5 mg raised mean hair count from 148 to 162 hairs (in a defined area), versus a modest rise from 144 to 150 in the placebo group.
- In the large Harcha et al. trial (over 900 participants), dutasteride 0.5 mg produced a mean increase of 90 hairs compared to 57 for finasteride 1 mg and a decrease of nearly 5 hairs for placebo.
- Olsen et al.’s dose-ranging RCT found dutasteride at 0.5 mg and 2.5 mg (mean increases of 95 and 110 hairs, respectively) significantly outperformed finasteride at 5 mg (76 hairs), while placebo declined by 32 hairs.
- Shanshanwal and Dhurat’s head-to-head RCT reported a mean increase of 23.8 hairs for dutasteride 0.5 mg versus only 4 hairs for finasteride 1 mg.
Lower doses of dutasteride (0.02 mg and 0.1 mg) also increased hair count significantly above placebo, though they did not consistently outperform finasteride.
Hair Thickness
Three studies examined hair thickness or diameter. Jung et al. found dutasteride 0.5 mg significantly improved hair thickness compared to finasteride 1 mg (mean diameter rising from 53 to 63 micrometers versus 52 to 53). Moftah et al. reported that 60.5% of dutasteride-treated female patients showed improved hair thickness, versus 22.5% in the placebo group. The Boersma et al. retrospective study found no significant difference in hair thickness between low-dose dutasteride (0.15 mg) and finasteride (1.25 mg) in women.
Adverse Events
Across all nine studies, adverse event profiles were comparable between dutasteride and finasteride. One trial (Jung et al.) reported a higher incidence of sexual dysfunction with dutasteride, but the large Harcha et al. trial found similar rates of adverse events and study withdrawals across all treatment groups, including placebo. The review authors concluded that the safety profiles of the two drugs were not meaningfully different.
Conclusions
This systematic review suggests that dutasteride, particularly at 0.5 mg daily, is more effective than finasteride at the standard 1 mg dose for increasing hair count and thickness in androgenetic alopecia. The data are strongest for men, where multiple RCTs converge on the same conclusion. For women, the evidence is encouraging but thin, resting on just two small studies with different designs and drug delivery methods.
Several important caveats deserve emphasis. Dutasteride is not FDA-approved for AGA, which means its use remains off-label. The total body of evidence here, nine studies, is modest, and study designs varied considerably. The longest follow-up in any included trial was roughly two to three years, so very long-term safety remains incompletely characterized. And while the review found comparable adverse events, the studies may have been underpowered to detect rare but serious side effects. The 1.57-fold increase in risk of sexual adverse effects reported in a separate meta-analysis of all 5-alpha reductase inhibitors is worth bearing in mind.
In short, the pharmacological rationale is clear: blocking more of the enzyme that produces DHT leads to more hair regrowth. But “more effective” does not mean “without trade-offs,” and individual risk-benefit conversations with a physician remain essential.
Relevance to Topical Alopecia Treatments
This review focused on oral formulations, but its findings have indirect implications for the growing field of topical DHT-pathway interventions. One of the included studies (Moftah et al.) used dutasteride delivered via mesotherapy, essentially injecting the drug directly into the scalp, and found significant improvements in both hair count and thickness in women. This raises an important question: can topical or locally delivered 5-alpha reductase inhibitors capture the superior efficacy of dutasteride while minimizing systemic exposure and its associated side effects?
Research into topical finasteride has shown promising early results in reducing scalp DHT with lower systemic absorption than oral dosing. Topical dutasteride formulations are also under investigation. For individuals concerned about the sexual or psychological side effects reported with oral 5-alpha reductase inhibitors, topical delivery may eventually offer a way to achieve meaningful DHT suppression at the follicle without the full systemic burden. However, no topical dutasteride product is currently approved for AGA, and the concentration, vehicle, and penetration characteristics matter enormously for efficacy.
Any comprehensive topical regimen for AGA, whether built around minoxidil, retinoids, or other active ingredients, benefits from understanding the DHT pathway that these oral drugs target. The superiority of dual 5-alpha reductase inhibition demonstrated here underscores just how central DHT suppression is to halting and reversing miniaturization.
Who This Study Matters Most For
- Men with AGA who have not responded adequately to finasteride: The data suggest dutasteride may offer additional benefit, and one included study specifically enrolled finasteride slow-responders who then improved on dutasteride.
- Women with pattern hair loss: Though evidence is limited, the two studies involving female participants showed meaningful improvements with dutasteride, particularly via mesotherapy. Women under 50 may benefit more from dutasteride while those over 50 may respond better to finasteride, according to one retrospective study, though this needs confirmation.
- Clinicians weighing off-label prescribing decisions: This review provides a concise summary of the comparative evidence that can inform, though not replace, individualized clinical judgment.
- Individuals researching topical alternatives: Anyone considering topical 5-alpha reductase inhibitors will find the oral efficacy data useful as a benchmark for what follicular DHT suppression can achieve.
Sources and Citations
Primary Study
Almudimeegh A, AlMutairi H, AlTassan F, AlQuraishi Y, Nagshabandi KN. Comparison between dutasteride and finasteride in hair regrowth and reversal of miniaturization in male and female androgenetic alopecia: a systematic review. Dermatology Reports. 2024;16:9909. doi:10.4081/dr.2024.9909
Selected References Cited by the Authors
- Zhou Z, Song S, Gao Z, et al. The efficacy and safety of dutasteride compared with finasteride in treating men with androgenetic alopecia: a systematic review and meta-analysis. Clin Interv Aging. 2019;14:399–406.
- Olsen EA, Hordinsky M, Whiting D, et al. The importance of dual 5α-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. J Am Acad Dermatol. 2006;55:1014–1023.
- Gubelin Harcha W, Barboza Martínez J, Tsai T-F, et al. A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. J Am Acad Dermatol. 2014;70:489–498.e3.
- Shanshanwal S, Dhurat R. Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled open-label, evaluator-blinded study. Indian J Dermatol Venereol Leprol. 2017;83:47.
- Moftah N, Moftah N, Abd-Elaziz G, et al. Mesotherapy using dutasteride-containing preparation in treatment of female pattern hair loss. J Eur Acad Dermatol Venereol. 2013;27:686–693.
- Lee S, Lee Y, Choe S, Lee W. Adverse sexual effects of treatment with finasteride or dutasteride for male androgenetic alopecia: a systematic review and meta-analysis. Acta Derm Venereol. 2018.